
Why the question?
I have had over a dozen questions in the last couple months about synthetic polyol esters and whether they form varnish or not. A couple examples of these questions below:
Hi Adam, I have had varnish issues in the past. My oil supplier is suggesting I switch to a POE to prevent this in future, is this correct, if so why isn’t everyone switching to these?
Question from UK
LearnOilAnalysis I have a question. Do you believe you can help? I read that neopolyol esters cannot form varnish, yet my filter company still thinks I should have a varnish removal unit on my injection moulding system. Am I being asked to buy something I do not require?
Question from Denmark
Hi, we have a [brand name omitted] VG 46 lubricated system and regular oil analysis including RULER and MPC. The data sheet says it’s a polyol ester yet I have an MPC rating of 18? Is this correct? The data sheet says it is very resistant to varnish and we have only had the oil in use for 2 years.
Question from Argentina
Hence I thought it’s about time I answered these questions as they have been coming from all across the world. To top it off I had a telephone question from a manufacturer of synthetic esters saying they had been having similar questions too regarding varnish potential on their products and setup a teams call to ask whether it was worth doing any varnish potential testing on these types of products.
Esters a quick recap of the basics
Ester based lubricants are nothing new. I’m not quite sure why this seems to be a hot topic lately, although with global oil prices very high I suspect a lot of attention has gone into biodegradable sources including esters. So with a lot of focus on these oils comes questions about their performance.
So what is an ester? In terms of the scope of this article it’s quite simple. It’s a reaction between an organic acid and alcohol. The reaction gives an ester plus water as a waste product.
The organic acids, usually fatty acids are normally derived from e.g. vegetable oils which are called triglycerides. For background of how you get from vegetable oil to acids, Triglycerides can be split into 3 fatty acids and one glycerol molecule. The fatty acids can then go to manufacture of an ester. To read a bit more about these processes and the chemistry you may enjoy my biodiesel article which covers much of the same chemistry and (spoiler alert) will also reveal the big weakness of esters too.
So why are esters good at resisting varnish?
Varnish is caused by oxidative processes on the oil to make acidic products and free radicals that form sludges. However with esters, if you think about it logically they started as acids and so have already gone past that stage and so are difficult to oxidise further. This makes them very oxidation resistant products. On top of this, esters have good solvent properties so they tend to hold onto varnish quite well before it deposits. This is as an extra reason they don’t readily deposit varnish.
Is there anything extra I should know?
Many esters by their design come from renewable sources and are biodegradable. This is usually a plus for many businesses trying to reduce their carbon footprint.
So esters don’t form varnish, is this correct?
In short, No it’s not correct. There are a couple of websites and marketing literature saying they can’t, but this is simply not true. They are very resistant but not impervious to varnish formation. This is because all lubricants will degrade. There is no perfect lubricant and often the advantages of a product in one situation become their downfall in another. In this case it comes from a simple principle in organic chemistry – All reactions are reversible.
All reactions are reversible
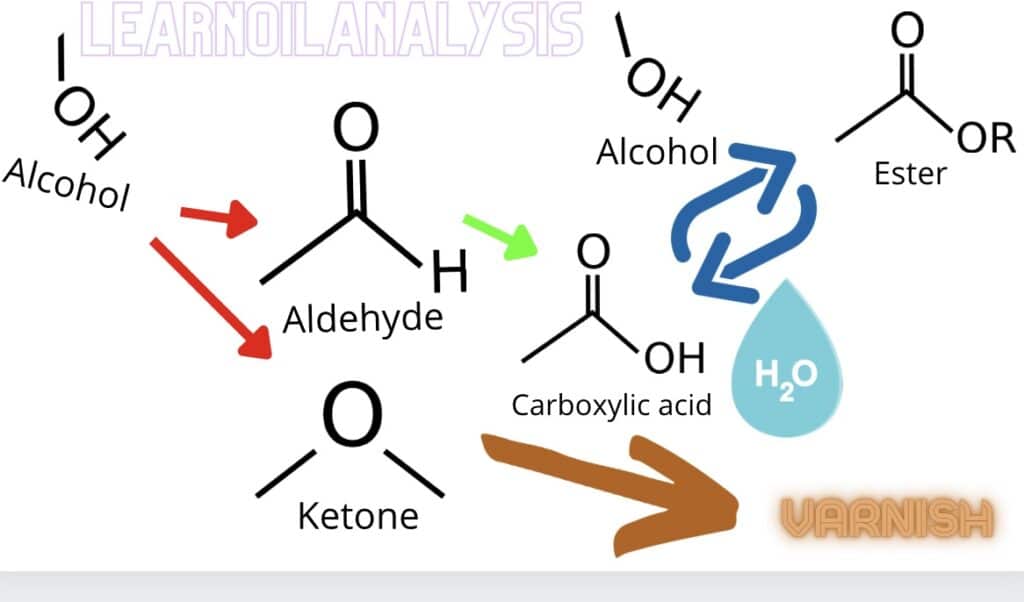
Chemistry of oxidation and varnish explained
The molecules we are interested in are aldehydes, ketones, carboxylic acids, alcohols and esters. The chemistry molecules above all show functional groups, ie. the important parts of a molecule. They are all depicted by a carbon backbone indicated by the black lines connected to significant elements. So a carboxylic acid is depicted as COOH as it’s a carbon with a double bond to O and a single bond to OH. The final bond goes to the next carbon in the chain. You don’t really need to understand too much chemistry to understand what’s going on. See the description below to see it’s very easy.
It all starts with an alcohol, not necessarily the drinking kind but the principle works with that too as wines age you want to avoid the acidic vinegar oxidation results. In this case depending on how the alcohol molecule is arranged it oxidises to an aldehyde or a ketone (red arrows). The aldehyde can further oxidise to a carboxylic acids (green arrow).
This acid can be combined with an alcohol to make an ester (blue arrow)
However the addition of water reverses the reaction and so you can end up with carboxylic acids which in turn can create sludges and deposits we term varnish.
Note for those organic chemists reading this please forgive the over simplification of the process but hopefully you will understand the purpose is for an introduction to varnish rather than a PHD thesis.
Organic chemistry is a little different to simple inorganic chemistry you learn in your first chemistry classes. You may have heard of acid + base gives salt plus water. In inorganic reactions the reactions are very violent and final so it takes a lot of energy to go backwards. However, the same isn’t true of organic reactions. They often want to go backwards or at least find some sort of equilibrium between the two sides of the reaction.
This is great with ester lubricants for their biodegradability as adding water will start converting them back to the fatty acids and alcohols they began as which can be further broken down in the environment over time.
However, this also means that water contamination in the system makes the oils very susceptible to hydrolysis. This is a process where they degrade in the presence of water to form their original precursors, which for esters are acidic and can cause corrosion and further degradation products and sludges which can be termed varnishes. So although the route may be slightly different the end result is the same.
Holding onto varnish may be a ticking time bomb
So I hope you now understand that varnish deposits can be produced by hydrolysis. This is not the only way as the oils also produce varnish by the typical free radical pathways, just this process tends to be slower as it is with most synthetics that tend to have good oxidation resistance.
Speaking of synthetics, typical group 3 and 4 base oils have worse solvent properties than the more basic minerals. This is because the refining / manufacturing process includes removal or synthesis without all the reactive areas of the molecules that are also good solvents. This is a known downfall of these lubricants as they struggle to hold onto anything in suspension including their additives at times. This is not a problem for esters which are naturally very good solvents and dissolve readily any oxidation byproducts in the oil. In fact many varnish removal chemicals use esters and good solvent property oils to help solubilise the varnish before an oil change.
This is good whilst the lubricant has good solvent properties, but over the long term varnish deposits can build up and agglomerate and when they do deposit when the oil is overwhelmed it occurs deep inside the system. Equally, a sudden shock of water contamination in an oil with lots of solubilised varnish can lead to large deposits of varnish within the system. So in essence this can be a ticking time bomb if not regularly tested for as part of a condition monitoring of lubricants package.
Summary – Test, Test and Test again
Ester based lubricants have many advantages over their mineral counterparts and many may choose to use them. They do have good performance properties in many areas but marketing material you may have seen saying these lubricants can’t ever form varnish and sludges is simply untrue. If you are being given advice that makes you believe you don’t need to test for the presence of varnish because you are using ester based oils you will likely be in for a nasty surprise. The only way you know how your oil is performing is by testing its condition and varnish potential is just one of those tests.
If you would like to find out about testing your oil then get in touch using the contact us button at the bottom right of this screen.
