
An interesting case the other day. A customer with a fleet of vans running on biodiesel suddenly started having problems with injectors failing. With any fuel system fault the first action is to take a fuel sample and many engine manufacturers won’t pay a warranty without a fuel sample to support the claim. When the sample arrived it was incredibly emulsified with a milky appearance.

Biodiesel according to the EN 14214 specification for new fuels should have no more than 500ppm of water (0.05%), but this sample contained around 25x more than it should do. The customer immediately called their bulk fuel supplier and suspected they were to blame for supplying poor quality wet fuel. However, the fuel supplier was able to provide their fuel batch certificate tested by an independent laboratory showing the fuel only contained 148ppm of water. So was one of the laboratories wrong and if so who was right as to the amount of water in the fuel? The answer was in fact both were right. The fuel indeed had very low water content at point of production, but by the time it had made it into the vehicles the water content had risen dramatically.
So why had the water risen so much?
To answer this question you need a very brief lesson on how biodiesels are made in a process called transesterification.
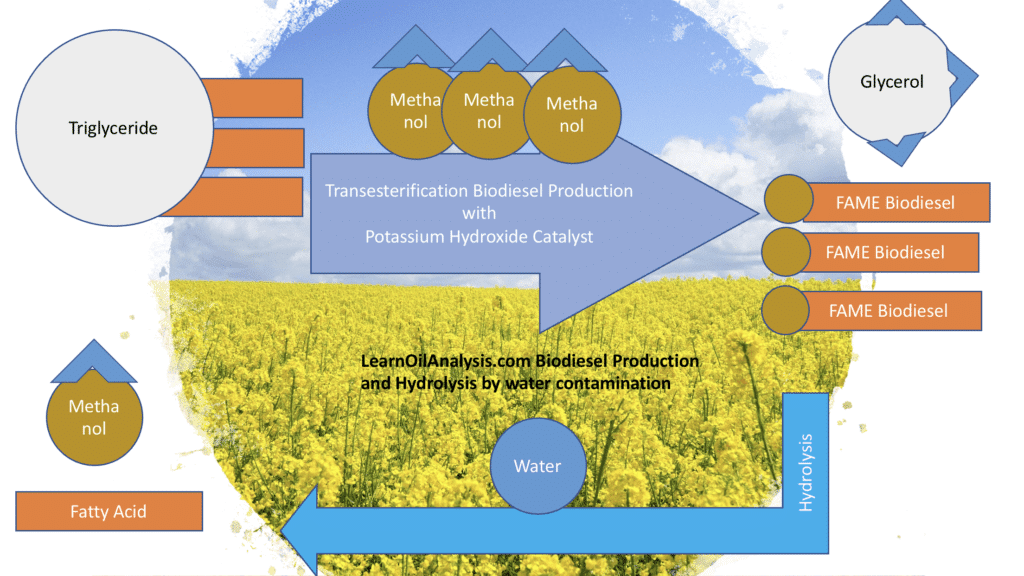
Biodiesel is made from vegetable or animal oils termed triglycerides, with rapeseed oil being a popular choice for biodiesel production. As can be seen in the diagram above the production process results in loss of water from the fatty acid part of the molecule as it forms the Ester. Now in layman’s terms the drying of the molecule means it acts a little like you if you had not had a drink all day. I.e. you would be desperate to get some water and if I put a bottle of water in front of you then you would probably guzzle the whole lot. In chemistry terms we say the reaction is reversible, meaning that it can go in the opposite direction to the condensation reaction and hydrolyse. So what determines the direction it goes in? This comes down to a simple principle called Le Chatelier’s principle which states that everything wants to be in equilibrium and if this is imbalanced it will try to return to this equilibrium state.
To explain this further, imagine you have two rooms which connect to each other at a very busy party. If you start off with everyone in one room that is overcrowded, people will start spilling over into the other room naturally. You can speed this up if you hired a bouncer (I.e. a catalyst) to direct people from one room to another. However eventually the second room will start getting over-crowded and people will start to want to go back to the first room until eventually there are roughly even numbers in both rooms. In production of biodiesel the presence of e.g. glycerol (I.e. people in the second room) slows down the reaction as the reaction wants to go back the other way. The only way to keep everyone entering the second room is to start removing people from the second room altogether (e.g. throwing out guests as they enter the second room) so the direction keeps going one way. Manufacturing facilities try to encourage the reaction to go one way by removing e.g. the glycerol as it builds up forcing it to keep going in one direction. So how does this all add up to water being a problem?
So how does water effect biodiesel?
Since the glycerol is removed from biodiesel production and so is the methanol, the reaction can’t go back the exact same way unless you add those back into the biodiesel. However, water contamination can start part of the reverse reaction turning the biodiesel back to fatty acids and methanol, but without glycerol the fatty acids never make the triglyceride stage, so you are just left with fatty acids, which are corrosive to the fuel system.
It doesn’t help that biodiesel naturally has a stronger affinity for water than petrochemical diesels. In fact about 30x more, meaning that water contamination in storage tanks needs to be well managed as biodiesel wants to absorb water 30x more than pure petrochemical diesels.
What is the effect of water on biodiesel?
Petrochemical diesels naturally shed the water so that it can be removed and are fairly chemically inert to the water as a result. The water itself can cause fuel system problems, but the diesel remains fairly much the same. This is part of the environmental reason for not wanting to use petrochemical diesels is that they do persist. Biodiesels in contrast are more biodegradable and readily react with water meaning their environmental impact is arguably much less than petrochemical diesels, but the biodegradability comes at the cost of their fragility. Hence, extra precautions need to be made to keep the fuel dry. Prevention is the main solution rather than curing water after ingress as the water chemically changes the biodiesel so fatty acids remain even after water removal.
How to make sure your fuel stays dry?
If you are bulk storing fuel then your fuel/tank supplier will be able to give you specifics for your situation, but common preventative measures include tank design to reduce water ingress through breathers and entry/inspection points. Use of desiccated breathers to ensure dry air enters the tank and regular draining of tank bottoms to reduce water build up all help to prevent excess water build up in the system. To check all is well though, you really should be sampling your fuel storage tanks regularly. Start with the most critical and bulk tanks first and these should be sampled quarterly or monthly and less critical tanks / vehicle system tanks 6 monthly or quarterly. Depending on the size of your tanks you should take multiple sampling locations including top, middle and bottom, or middle and bottom on smaller tanks.
How do I start sampling my fuel?
To find out more about how to sample your machinery and storage tanks please get in touch with the contact us button at the bottom right of this screen.
