I was asked the question the other day how high would silicon have to be in an oil before you would change it based on that parameter alone. The system in question was an injection moulding machine which have exceptionally strict cleanliness levels. The samples were not tested by our lab, but the lab they were using was a reputable one and the results for silicon were between 40 and 70ppm with nothing else highlighted. To answer this question, it very much depends on the source of Silicon as some are actually useful and some are very damaging to the system. Annoyingly they all have the same primary chemical element of Silicon so it takes a little experience to spot which one it might be.
You can also watch a video summary of this article if reading seems like too much effort.
So what are the sources of Silicon and how do I fix them if they need fixing?
Silicon is the second most abundant element on earth after Oxygen, so it can be found in lots of places. Silicon has a few sources namely:
- Silicon particles of sand. Sand is silicon dioxide and can be found pretty much everywhere on earth. Even the finest particles are about 70 microns in diameter and are very abrasive in the system. Sand can be an airborne contaminant depending on the local environment around the machinery. This is usually fairly simple to remove by filtration of the oil and breathers. It can also be casting debris not fully removed on newly manufactured components on initial oil fills. The corrective actions are usually changing the oil or filtration assuming the sample was not contaminated at sampling.
- Silicon as environmental dirt – this is probably the most common and easily recognisable source of silicon in your oil. Aluminium silicates are what makes up rocks and clays. The classic identification of dirt is silicon and aluminium in a 2:1 or 3:1 ratio. This is usually corrected with filtration / oil changes and investigating the source such as breathers, poor sealing, air intakes, dirty oil storage containers and a dirty sampling point etc.
- Silicon as part of the steel wear material – most steels contain silicon (usually as silicon carbide) as it helps as a deoxidiser (ie in removing oxygen from the ore) to strengthen the steel. It can be found up to 1% but it can be higher for welding materials or electrical uses. It doesn’t generally get commented on in terms of oil analysis as the iron is a much stronger indicator of wear (over 100 times the value of the silicon) and corrective actions are made before silicon becomes significant. So high silicon is usually in an oil analysis sample going to be one of the other reasons unless there is phenomenally high iron wear.
- Silicon as ceramic material wear – this really needs prior knowledge of your system make-up but high silicon on its own can be wear if for instance you have a component made up of very durable ceramic materials such as silicon carbide commonly used in e.g. ceramic brake materials. However since the hardness is about 4x that of steel it is quite rare to see Silicon as the predominant wear metal.
- Silicon as sand or as siloxanes – If you work in landfill you will know silicon is the bane of your life in terms of problems with the engine. Silicon has a couple sources within this system, but one is far more likely the source than the other. Let’s start with the minor source first and that is from sand particulate entry through gas supply. To deter bad odours coming from the landfill, loss of methane gas to the atmosphere and rats etc from entering the landfills the rubbish/trash is capped/covered with multiple layers, one of which includes sand. Sand and the other layers of this capping system are used to assist in drainage and prevent harmful chemicals entering the surrounding environment. If there is poor filtration of the gas abrasive particles of sand can be carried into the system and can make their way into the engine where they cause wear. This is generally solvable in system design and gas filtration. However, the much bigger problem is sand from siloxanes. Siloxanes are found in many household products such as cosmetics and when these break down at landfill the chemically lighter parts form part of the landfill gas that enters the engine. These are not a problem by themselves, but when they enter the engine and are burnt they are converted to silicon dioxide (sand), which is very abrasive and causes wear. The solving of high siloxanes tends to be very difficult and although some aftermarket filtration systems are available most landfill operators still need to use oil changes to reduce the build up of silicon in their oils. Hence these tend to be some of the most regular oil samplers; sometimes having to change their oils weekly. The important thing to note is the effect of silicon in terms of is it still in the siloxane form or an abrasive form such as sand when detected. The LubeWear technique of monitoring wear metals at the very earliest stages has had success in allowing some operators to extend drain intervals by monitoring for wear rather than solely on silicon alone as the point to change the oil. Naturally not missing scheduled samples and quickly reacting to lab data becomes critical in this type of extension.
- Silicon as silicone oil/anti-foam – silicone oil can be used as a lubricant, so this is assuming your lubricant is not silicone based, in which if it is then the amount of silicon doesn’t matter. However, silicone oil is also an additive used between 5 and 30ppm in lubricating oils to reduce foaming. The higher levels tend to be used in more prone to foaming lubricants such as engine oils that have high degrees of detergent additives, but can also be found in many hydraulic, compressor, gear and turbine oils too. Higher levels can be found based on sampling when the lubricant is not fully mixed. This is because by design antifoam additive is not dissolved/suspended easily in the lubricant (see explanation of how antifoam works below) as this makes it a good anti-foam compound. The antifoam is more dense than the lubricant and can sink to the bottom of the system over time when not under constant agitation of the machinery. This is a common issue with machinery that is stationary before sampling such as wind turbines where sampling is done during low wind speeds and when samples are taken from IBCs that have been standing for long periods before drawing oil from near the bottom. In these cases antifoam can quite quickly drop out of the oil and can be artificially high in a sample if taken near the bottom. It is also worth noting that nearly every filtration system will remove silicone oil over time and so it is not uncommon to find silicone oil in filter reports. You can also see foaming after a filter change as the silicone oil in the filters that collapsed foam as it passed through is removed. Overall, silicone oil is not a problem in your system unless it is at such a level it is diluting additives or altering the viscosity. Otherwise it is just part of your oils natural anti-foam package. If you see high silicon and wear, then this is a different matter as it could be abrasive silicon or it could be silicone oil to such a high degree it is causing high air entrainment so the bubbles can’t be released which can lead to increased wear in the system as any entrained air is a bad thing for your system. As with any additive it is important to get the right balance for your system. This is particularly important if you are using supplementary antifoam additives to fix foaming issues.
- Silicon as silicone grease / sealant – silicone grease is a common grease used in many systems. The most common reasons for this being in the main lubricating oil are because (a) a new component has been fitted containing grease/re-greased such as a pump and this has leaked into the oil; or (b) grease has been used to try seal an oil leak. The former usually suggests the system has been over-greased before fitting/sealing issue whilst the latter is a bad practice for sealing oil leaks and can lead to considerable wear. If this is found the system should be closely monitored for physical changes such as increased viscosity and increased wear. The general fix to this type of problem is an oil change, but depending on the volumes involved and cost of loss of production then the end user may choose to delay the change and just monitor for wear more regularly to extend the oil life.
bursting.
What is foam and how does anti-foam work?
To answer this we must first define what foam is. Foam is the gas dispersed in a liquid that when the gas bubbles reach the surface the bubbles do not burst faster than they are created. In a pure liquid it is thermodynamically unstable for this to happen. Hence there needs to be something else in the liquid to stabilise this foam production and this can be explained by the Marangoni effect. If you are into your wines you may already be aware of this term as the cause of the “tears of wine” effect. This nice YouTube video below gives a really good demonstration of this effect in a wine glass if you have never seen it.
You can also see these swirls that give the pretty colours to a soap bubble (see picture above).
The effect works because all the liquid molecules are slightly attracted to each other by very weak molecular forces. These combine to give surface tension.
How do these molecules produce surface tension?
If you imagine you are a molecule in the middle of a glass of water you are attracted equally to all the other molecules and so you stay in place. However if you were one near the top of the glass the overall direction of pull is down as there is no liquid above you to pull you up, which gives the effect we call surface tension.
If you imagine you are now an air bubble, trying to form a foam bubble, it’s a little like when you try blow a balloon at the start. If you recall, the very early part is the most difficult because the rubber film has too much tension before it is stretched.
In a pure compound the surface tension between the liquid and the air is very high much like that early stage of a balloon blowing making it difficult to separate the liquid molecules to form a bubble. The addition of surfactants lower the surface tension so the surface is much easier to overcome and so much easier to form a bubble.
How does a surfactant work and how is foam formed?
The surfactant molecules work as they are equally attracted to both of the surface tension phases ie the liquid and gas, and by the Marangoni effect they concentrate at areas of high surface tension, dragging more liquid with them and thickening the liquid film. You may think this would create more tension as you are thickening the film, but instead it stabilises that area making it stronger and resistant to bursting where the bubble might otherwise be at risk of collapsing. Think of it like adding a reinforced lintel to a doorway to stop it collapsing under the weight of the house above. So this extra material goes and reinforces the areas of the bubble that might otherwise collapse under the ‘weight’ of surface tension.
How does antifoam work?
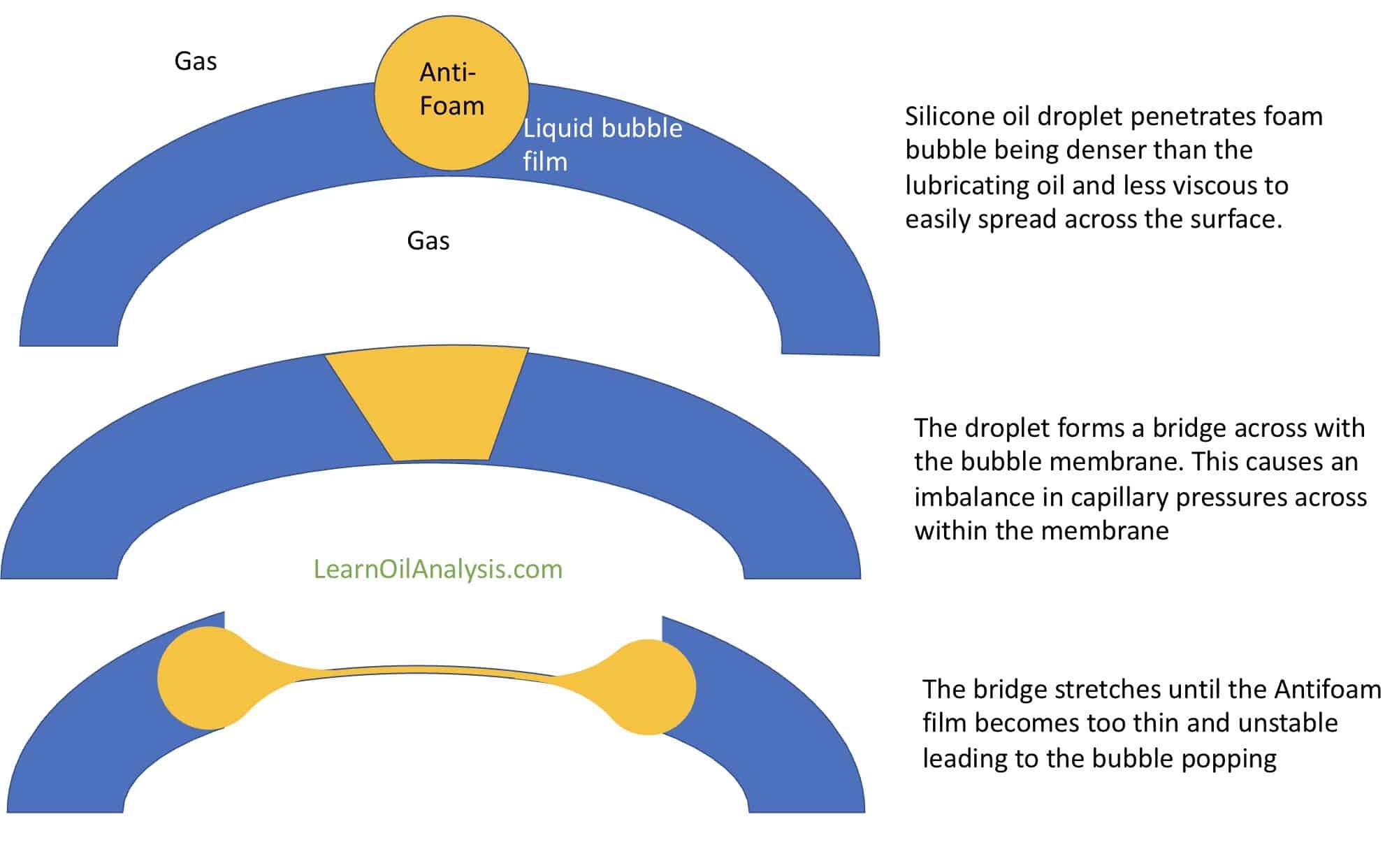
Antifoam works by disrupting the effects of surfactants and other products that are reducing surface tension, making the bubbles less elastic and more stiff/rigid – hence easier to break. To do this they firstly need to be very low viscosity to allow easy movement. Secondly they need to not be soluble in the main fluid. They also often need to be denser to penetrate the foam and the main liquid. Silicone oils density is higher than that of most mineral lubricating oils which also helps with its antifoam properties.
Anti-foams replace the surface active molecules that are stabilising the foam. They do this by bridging the gap across the foam bubble liquid film and cause a weak spot between bubbles. This causes foam bubbles to rupture forming bigger and bigger bubbles (as 2 bubbles become 1 big bubble) that eventually burst altogether. The diagram below helps explain this bridging effect.
What to do about high silicon?
Your individual report will advice you on some practical next steps if any are needed, but as a general rule if silicon is the only flag then just continue monitoring. It’s only if it’s causing other parameters to be abnormal or increased wear you would start to address the problem.
If you would like more help on how to handle a silicon issue in your system then get in touch to discuss with us by clicking the contact us button on the bottom right.