The average sample that comes into the lab has around 30 to 60 different tests on it depending on the sample type and suite. However, labs can’t always perform every one of them every time. This is sometimes because the customer hasn’t provided enough sample so if you have a test that requires 50 ml and the customer only provided 30ml then the lab will naturally struggle to perform the test. However, today I want to talk about when a lab chooses not to perform a test because a sample is too contaminated. There are a few reasons your sample will not have a test performed and I will include a couple of pictures to explain as well. These are all very rare scenarios, but if you send lots of samples then eventually you may see one of these situations.
Why won’t the lab perform a test all the time regardless of the contamination level?
It may seem strange at first that if you can do something that the lab chooses not to in some circumstances. I do know some labs will perform a test regardless of contamination level as it takes time and effort to treat one sample differently. It is easier to do the test than not to. This is a dangerous approach to take though as by the act of producing a result and displaying it on the report, the customer will automatically assume that result is accurate when in reality it may not be. Sure the calibrations and quality control standards are all fine, but that does not mean this individual sample is, which may seem strange so let me give you an example.
Case Study 1 – Viscosity of a gear oil
Let’s say you have a sample of gear oil that is supposed to be an ISO VG 220 from a wind turbine. Your quality control sample also happens to be an ISO VG 220 too. This is a perfect comparison as your quality control standard is in the same region as your sample, so you can assume if your quality control sample passes the result for the gear sample is very accurate.
The lab runs the sample and gets a result of 334 mm2/s. This seems strange, but you also have a quality control sample of a 320 ISO VG (the other commonly used wind turbine gear oil grade) and that passes perfectly too. Do you quote the result? Many labs would at this stage because the QCs (quality controls) passed. I would personally re-run the sample to make sure. Let’s say the result came out at 174 mm2/s now. So we run again and get 194 mm/s. Which result is right? In scientific terms, you should average all 3 to get 234 mm2/s meaning the result actually passes for the sample (within +/- 10% of the 220 value). The customer sees on their report the viscosity of 234mms/2 at 40 degrees C and thinks all is ok. However, it is not.
Those who have seen this sort of thing already may be thinking there is going to be lots of water in the sample causing the problem result. However, the water was <0.1% and was normal. The sample was dark, but no other obvious cause such as oxidation of the sample.
The lab technician reported the value as unable to obtain an accurate result rather than taking the average or simply taking the first result.
When the diagnostician looked at the data, the only thing that identified this problem was the fact we could not get a reproducible viscosity. We did some extra investigation and used a very bright light behind the sample, in which you can see there is actually a layering of the sample (note the dark layer just above the cog symbol).
It turned out the layer at the top of the sample was 320 mineral gear oil and the layer at the bottom was 220 synthetic gear oil. The two products were not compatible and caused the problem viscosity results. It turned out the gearbox had been switched from a mineral 320 to a synthetic 320, 6 months earlier.
So why don’t we just quote every result regardless?
Put simply even though you may be able to pipette some sample into lab instrument and get a result, this doesn’t mean the result will have any value to put it on the report. The experienced lab team will use their discretion as to whether a result should go onto the report and will highlight where a sample is too contaminated to perform particular tests.
I will now go through some of the most common reasons results will not be provided.
Viscosity – when the sample boils
Viscosity is the most important property of lubricants, so to get a report without it may seem strange. I already gave an example with incompatibility of products. However, another common reason to not perform the test is heavy water contamination in which particularly at 100’C measurements causes the water to boil and gives you an inaccurate result.
So the lab is left with a choice of what to do. Some will simply quote the result of the sample that has boiled in the viscometer tube. The very fact the sample has boiled means the values will be meaningless and no better than a random number.
Some labs will choose to sample the top oil layer or even boil off the water first to remove the effect of the water, but these bias towards favourable results by ignoring the contamination and give a false sense of security. The water effect is not being ignored by your machinery, so why should we ignore it during the testing. Hence why I think it is better to highlight the sample is too contaminated to perform the test to prompt the customer to address the issue.
Particle counts – when it is too dirty?
I know quite a few filtration industry professionals will consider an ISO4406 particle count being performed on oil samples as sacrosanct. For many is the single biggest parameter they will use to make filtration decisions. So please be patient whilst I explain there are some circumstances performing a particle count are not appropriate.
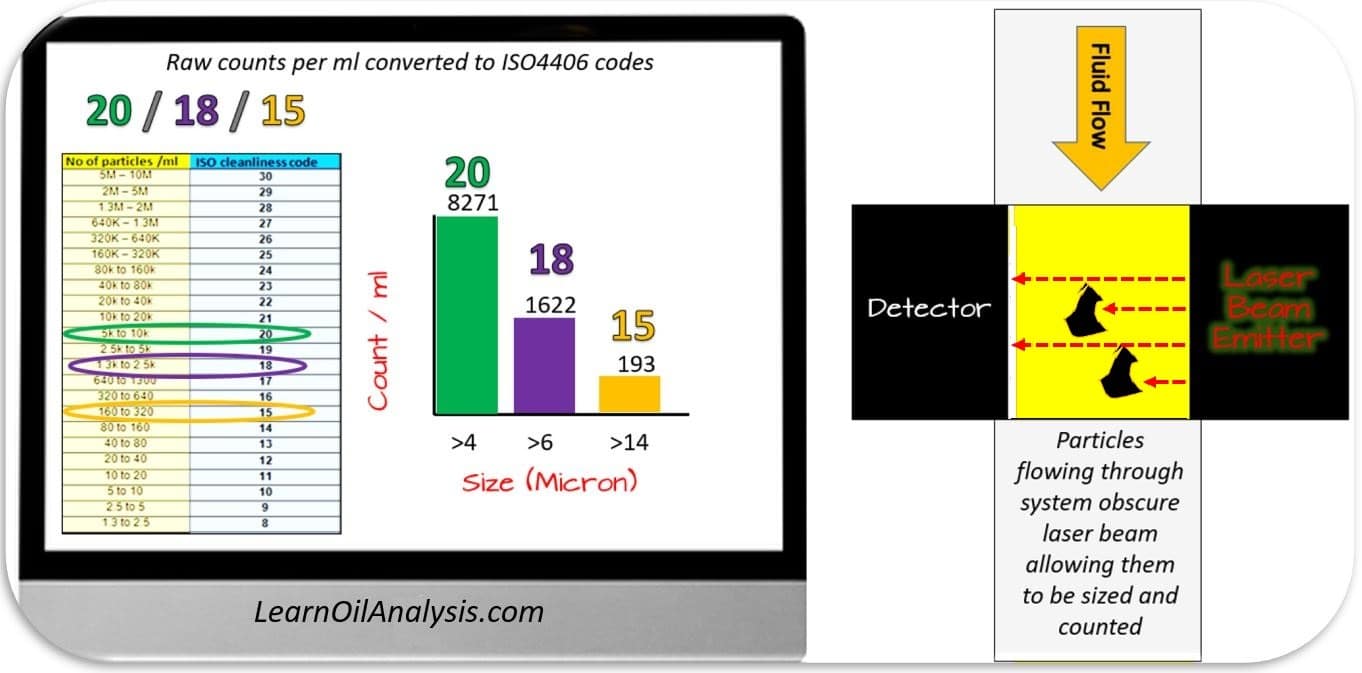
The classic way of measuring the number of particles in a sample is through laser particle counting. As you will see in the diagram below the fluid passes through a laser detector that measures the number of particles as they block the light beams.
Particle counters will detect anything that blocks or deflects the light which includes:
- Hard particles such as dirt and small wear particles
- Soft particles such as water, oxidised oil and varnish, air bubbles and not fully solubilized oil additives e.g. silicone oil anti-foam.
Particle counters are very sensitive instruments designed to look at very low levels of contamination in samples and hence if a sample is visibly contaminated then it is likely too dirty to process.
Why is the oil too dirty to get a particle count?
I do get asked this whenever we have had to cancel a particle count “can’t you just make an exception for this sample”. To explain this I first need to explain why we don’t perform the test and it’s not to be awkward. Since a particle counter is designed to test the cleanliness of fluids, it means the particle counter itself is very susceptible to contamination. Running grossly contaminated samples repeatedly through the counter means that over time the accuracy of the counter (even with annual re-calibrations and regular quality controls) will fall and usually results in overly high counts in the long term. I have worked with quite a few labs over the years and this is a very common problem.
In the short term, it will also cause very high counts for the next few samples through the “carryover” effect. Any respectable lab will clean the instrument between samples, usually with the next sample to be measured to avoid counting the solvent wash cleanliness. However, each clean only removes about 90% of the contamination. So two flushes should do 99% and 3 does 99.9% in a perfect world. In reality, it is never this good, but let’s assume you have removed 95% with a 3 wash cycle. That still leaves 5% in the next sample to be potentially counted. The lab can have quality control samples in the batch (most labs usually use 1 in 20 samples as quality control). Let us assume you have 20 samples and a QC at each end of the batch. If sample 5 was grossly contaminated, by the time the QC has been reached the contamination seen in samples 6, 7 and 8 is no longer visible giving a false pass. Our lab operates if a sample has an overly high particle count this automatically triggers an extra wash cycle and a QC until the QC passes again, which usually addresses this problem, but for labs who do not have spare capacity on their particle counters for extra quality control, they cannot afford the time for these extra checks. I have known a sample to take 3 hours of continuous flushing after running (the test usually takes less than 3 minutes) to clean it after accidentally running a very dirty sample.
So how do labs address the contaminated sample issue?
Some labs I know operate a zero-tolerance strategy of if there is any visible contamination e.g. 1 spec of dirt in an otherwise clear and bright sample they don’t process for particle count, which although addresses the issue doesn’t understand the practicalities of having to sample in perhaps quite dirty operating conditions.
I personally do allow a few visible specks of debris through to accommodate less than perfect sampling technique, but draw the line at a load of gravel at the bottom of the bottle. I personally tell my staff that if there is too much dirt to count at the bottom of the bottle or if a sample were a drink (don’t drink any samples please) and you wouldn’t drink it then don’t put it through the particle counter.
So how do labs get round the problem of too dirty samples for a particle count?
Some labs will just run everything anyway, and over-time the quality of results their counter produces will diminish until the next calibration. Many labs now use solvent dilution as it eases automation of particle counting systems and removes a lot of the awkward softer particles such as water. If a sample is grossly contaminated you just increase the dilution until you get to something you would put through the counter. The issue with this though is when you multiply the results by the dilution to bring it back to the original results you also multiply your errors meaning that you can get quite inaccurate data.
Equally, solvent diluting by discounting some of the contaminants gives a false sense of security. If you have a sample with 1% water, removing it by solvent dilution falsely gives a better result than they should be alerted.
Other labs just run the supernatant (I.e. the top of the bottle) but this again ignores all contamination at the bottom.
Finally, another solution is to do a patch, which is to pass the sample through a filter patch paper and provide a patch scan or a visual patch count. These by the nature of testing will ignore anything that passes through the filter patch and give much lower counts than a laser count would. This again gives a false sense of security.
Case study 2 – When a particle count is mandatory?
I know a few companies that to comply with their own company policies they need an ISO particle count. This is great in practice and seems a very top-down mentality. A paper factory I work with has 460 gear oils was told they needed to get particle counts on all their gears when they had a consultant lubrication engineer from the USA come in and say that all gearboxes must be better than 18/16/13 (8 times cleaner than their new oil from the barrel). I personally don’t believe in doing cleanliness codes on gear oils as unless you have a filtration system to address a high result you simply just generate a number in which the customer has no mechanism to resolve. In reality, the ISO 4406 limit this low would also cause false alarms and distractions from the issues such as high water and wear the client was experiencing (paper factories are very wet environments). After much discussion, we agreed only to add the test to any systems only when filtration was fitted.
Another company in the aviation manufacturing industry had mandatory particle count testing as company policy. Unfortunately, the very first batch we received, the samples were grossly contaminated with a mix of mineral and synthetic fluid (as the customer used the same offline filter cart for all machines on-site). This sample below was taken from the top of the customer’s tank. Believe it or not, the bit that is the pink at the bottom is actually the right synthetic oil and the stuff on the top is the contamination. This meant on their samples we couldn’t provide an ISO code, which the customer was at first unhappy about, but once we explained why, they were able to address the issue. Although we could have theoretically provided a result provided by the particle counter or done a visual patch, the motivator of the sample being too contaminated to test meant they acted to resolve the problem quicker so they could get the code for their audit at the end of the year. The customer replaced the filter carts so they had one for mineral and one for synthetic on-site and replaced the oil on 2 machines.
Below is the sample from the refill of the system and yes the customer got their particle count and passed the audit.
If you would like to find out more about our test suites and how you can get the most value from them please use the contact us button below.