An interesting question today from a customer regarding how oils degrade and I thought I would make an article to answer.
“Hi Learn Oil Analysis,
I have heard lots of conflicting information about what is the best way to test how degraded an oil is. Some say its measuring varnish potential, others measuring RULER and others RPVOT. Which one should I use?
Hope you can help 🙂
Tim”
This is a great question as being a lab owner myself everyone is constantly trying to sell you their new way to test oil and all claim to be the silver bullet to highlight all issues. Some of these are great, but ultimately it can often be a combination or a matter of timing and what you want to achieve. So firstly lets cover what is a lubricant made up of?
What does a lubricant contain that we are looking to measure?
Lubricating oils are made up of two main constituents, namely the base oil and the additives. In terms of additives, we are interested in anti-oxidants which are usually phenols, amines or anti-wear based or a combination of all 3. For a lubricant to form sludges and varnishes which can cause problems in your machines both the anti-oxidant additives and the lubricant base oil natural oxidation-resistant properties need to be overwhelmed.
With additives, the more of these additives present the more anti-oxidant properties the oil will have, but some combination additives have a recycling effect to rejuvenate the anti-oxidant over time hence it is not a simple case of the more the better in every single instance as some combinations of additives may be better than the sum of their individual parts.
With base oil, taking petroleum-based oils which is the vast majority still of lubricants these are divided into different classes generally groups 1 to 4. The higher the group the more highly refined or in the case of group 4 the more highly synthesised the lubricant meaning the parts of the lubricant molecules that make them susceptible to oxidation have been largely removed such as double bonds and none carbon-hydrogen and carbon-carbon bonds. Without going too much into the chemistry it is easier to think for groups 1 to 3 the higher up the groups you go the more weak links you are taking out of the lubricant in the defences against oxidation, wherewith group 4 you just build the defences without weak links in the first place.
What is needed for a lubricant to degrade?
This is a very big topic and much research is done into this with millions invested to try to make lubricants last longer, but I will keep this brief for the purposes of the explanation below.
Lubricants degrade oxidatively through 3 sources namely oxygen, heat and a catalyst such as iron.
- Oxygen – Oxygen is part of the air we breathe so reducing its presence in the machinery is exceptionally difficult even in sealed systems as the head of tanks contains air and so does lubricant storage etc.
- Heat – For every 10’C we increase the temperature we nominally half the life of the oil. So if you have two machines with one running at 40’C and another at 60’C this means the one running at 60’C with all other things being equal will have only 1/4 the life of the other machine (half and then half again for each 10’C rise).
- Catalysts – the machinery will often be made of the catalyst materials such as iron, hence this usually cannot be avoided, but high wear rates greatly increase the surface area of the fresh surface metal that has not been inactivated by anti-wear additives and will increase oxidation.
- Water – acts as a medium for the oxidation reactions to take place and allow ions to be stabilised during their intermediary steps in the reaction. These also hinder and strip the lubricant of protective additives.
How does a lubricant degrade and how to test for it?
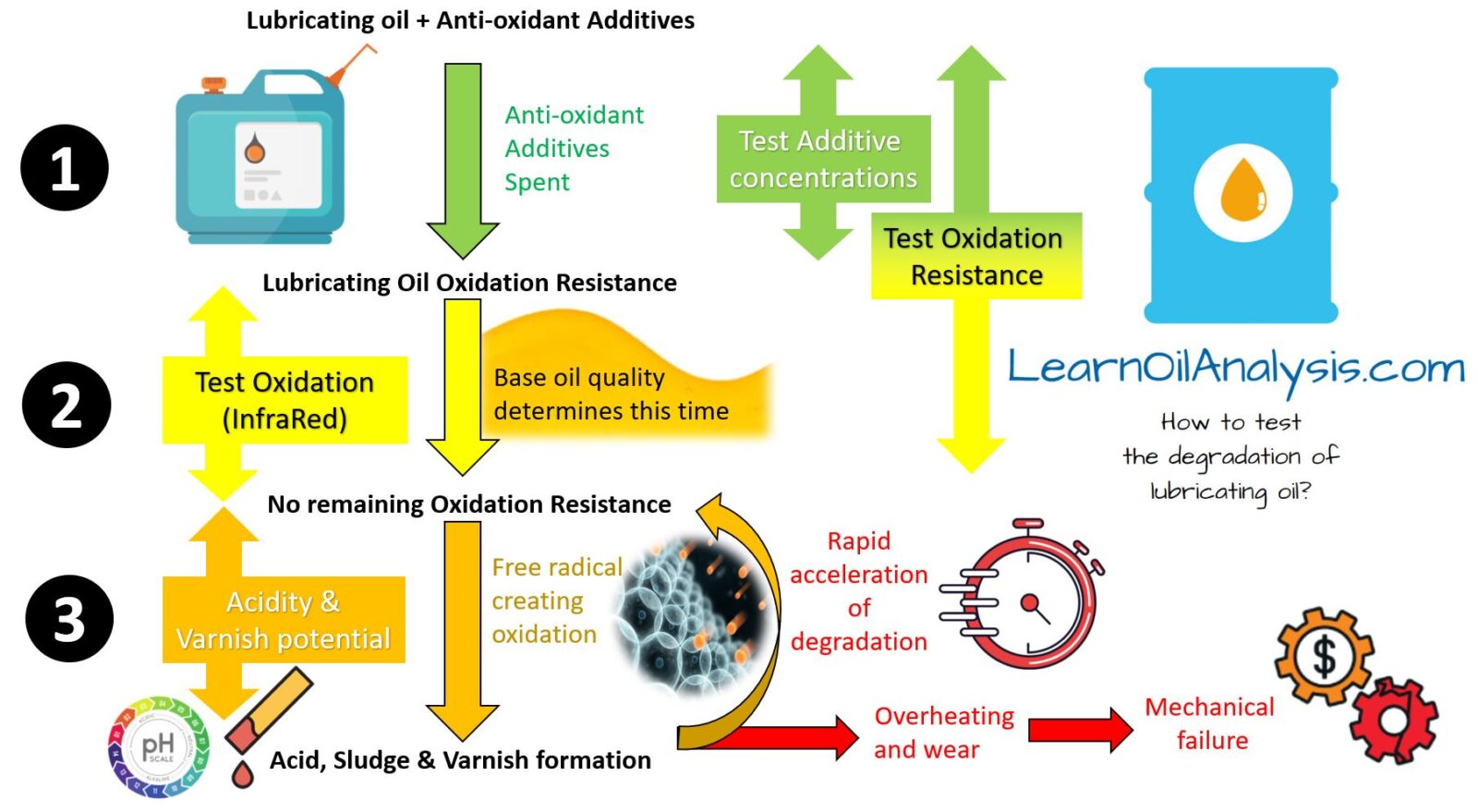
Please have a read of the diagram below whilst reading through these steps to assist in putting this into context.
Step 1 – Measure Anti-oxidant concentrations Directly
Step one involves the anti-oxidant additives being spent. This can sometimes be seen with a simple colour change or darkening of the lubricant as many phenols are used as dyes and change colour as they react. However, very mild changes in additive concentrations can cause huge changes in colour and you can have very similar colours with massively different additive concentrations. Colour can be tested using the ASTM colour scale.
Colour Advantage: quick and cheap to perform.
Colour disadvantage: level of colour change not always related to level of depletion.
In addition to colour, measurement of individual additive concentrations can be used with popular methods including the linear sweep voltammetry testing (RULER test is the most commonly known brand but other players have now entered the market with more modern equivalents), which measures as a percentage the amount of anti-oxidant vs a new oil. This is relatively quick to perform compared to indirect measurement (see RPVOT later in the article) in a lab and has a good reproducibility (I.e. error of the test) of around +/- 10% compared to RPVOT reproducibility of +- 22%
The limitation of this method is that it assumes customers don’t mix brands of oils, which for large turbine systems customers may do a 25% oil change each year because of budget reasons rather than any diagnostic benefit rather than a full oil change. Hence for these situations, it is not ideal as you may have a mix of brands of oils. It can still be used, but you are only comparing life to the current fill rather than the original oil if mixing or unsure of brands.
Linear sweep advantage: quick (results in under an hour at a lab), low oil volume of only a few ml required, can now be automated with more precise sensor technology these days and generally depletes earlier than other measurements. So it is good if you want to air on the side of caution as it will alert far earlier than alternatives.
Linear sweep Disadvantage: older manual forms of the technology and methods not very good with non-perfect oil additive blends. Only measures the additives and not the base oil degradation.
Step 2 – Measure Oxidation
Measuring oxidation by infra-red analysis identifies the lubricant beginning to oxidise. This step may be a very short or very long stage depending on the base oil quality, with the higher quality base oils lasting for longer. Hence, simply changing when the anti-oxidants are spent can be a false economy, especially when using highly refined or synthetic lubricants that may have plenty of life left. Oxidation is a good indicator of the base oil beginning to degrade but does have interferences with certain additives giving an artificially high result when e.g. ester additives are present.
Advantage: can be performed routinely. Measures base oil degradation well.
Disadvantage: certain carbonyl-containing additives can give false positives and technology is not sensitive to small degradation changes to make a difference.
Step 1 & 2 combination – Measure Oxidation Resistance
This is commonly performed by the RPVOT aka RBOT (Rotating Pressure Vessel Oxidation Test), which involves taking a sample of the oil in combination with a catalyst and pure oxygen under pressure to attempt to artificially and rapidly degrade a lubricant. The test is measured in minutes with a common cut of >500 minutes being a good pass result and <100 minutes being considered a poor result. It could be argued this test is a truer reflection of lubricant degradation than antioxidant methods because it is measuring both the anti-oxidant and base oil resistances together. Although the diagram shows additives spent first then the base oil, in reality it is both being degraded the entire time, but predominantly the antioxidant additives at the beginning and as this protection is removed it then includes the base oil. RPVOT has been the gold standard test in the industry for many decades now but does increase the sample price to cover the high consumables such as the copper catalyst coils that are used with each test. However, in recent years there has been an increase in additives that nullify the effects of the RPVOT test by deactivating the copper catalysts and giving artificially high RPVOT values even when the lubricant is severely degraded. This has caused a shift change in alarm limits, but also because not everyone is using these specialist additives is meaning there was until recently a two-tier system in measurement and that is why our laboratory developed some new measurement techniques to discount for the effect of these falsely good condition results as a side effect of adding additives not traditionally viewed as anti-oxidants that give an artificial anti-oxidant effect.
How does the RPVOT work in detail?
The Rotating Pressure Vessel Oxidation Test (RPVOT, aka RBOT) uses an oxygen pressured vessel to evaluate the oxidation stability of new and used oils with water and a copper catalyst coil at 140°C or 150°C depending on the variation of the method used. The oil is electrically heated in a dry pressurised bath with pure oxygen and the sample rotates axially at 100 RPM at a stable pressure of approximately 90 psi. Hence we have all the ideal conditions for oxidation with a copper catalyst, heat, water and pure oxygen. As the oil oxidation resistance of the base oil and additives is overcome the oxygen forms part of the lubricant molecules and hence is no longer exerting a pressure as a gas. This drop in pressure below a threshold as dictated by the method is considered the end of the test and the resistance is considered to have been overcome.
Advantage of RPVOT: gold standard test. Not brand specific. Measures base oil and additive depletion.
Disadvantage RPVOT: less accurate (22% reproducibility) compared to linear sweep testing at (10%). Test is slow and expensive. Because it measures both additives and base oils it will generally drop much later than linear sweep testing. It is possible to cheat the test with selection of additives used by the manufacturer to give an artificially high “good” result, but OAL has managed to correct for this problem in their analysis.
Step 3 – Acidity and varnish
At this stage, the oil may be beyond salvage as the oil begins to chemically break down forming free radicals that rapidly increase the rate of oxidation. Hence things can change very quickly at this part of the process and decisions may need to be made rapidly to prevent failures. One of the products is the varnish which can block, coat and seize components and also insulates the system to prevent heat loss, which also increases the rate of degradation. This eventually leads to wear increases from the more acidic environment and poor lubricant properties of sludged oil, which is also a catalyst for degradation. Hence you can see how everything is feeding back into the process to generate further degradation at this stage and the end result is a failure. This can be e.g. a seized component, blocked filters, system overheating or other serious mechanical failures. Hence this is the stage you often want to avoid.
Advantages: Gives a good indicator of the likelihood of the oil to varnish and the level of acidity of the oil currently. Usually occur quite late in the degradation process so unusual to get a false alarm if baseline values are known.
Disadvantage: If oil is actively depositing varnish or developing high acidity problems can already be occurring, so this is a late-stage fault and needs potentially fast actions to take place.
Which is the best test?
There is no “BEST” test in this regard for all scenarios and although many labs will have preferences of methods to use it depends on your individual situation and what you are trying to achieve. If you are in the middle of nowhere and have no access to a lab and don’t care how your oil compares to other oils then something like a remaining useful life or another portable voltammetry tester would be ideal as they are designed for field use and are very quick and simple to perform for non-lab people.
If you want to get a top to bottom full health check assessment of your oil’s level of degradation then you may choose to go for instance, for a more modern Remaining Useful Life voltammetry lab test with lab levels of precision. This combined with Oil Analysis Laboratories anti-oxidant test plus an RPVOT and a varnish potential will cover all the bases for you. It all really depends on the criticality of your machinery and what you are trying to achieve. If you are unsure what you need why not use the contact us button at the bottom right of this screen and get in touch explaining a bit about your machinery so we can best advise you what tests would help identify the degradation of your oil. We will also be able to help you with detecting the wear and contamination of your oil which are also very important aspects to measure in any lube analysis programme.