I had a question asked to my blog recently. The question was as follows.
“I have heard of tests such as TAN, TBN and oxidation, but what is the best way to tell how degraded my lubricant is. Do the tests all do the same thing?”
James – lube and filtration manager

Hi James, yes the various tests have all different uses. I will not cover contamination causes such as e.g. water that degrade the lubricant and work more on oxidative reasons in this explanation. To explain let’s first understand how lubricants degrade.
What are the different ways of testing lubricant degradation?
These can be grouped into 4 categories based on the stage of the process: (1) Additive depletion, (2)oxidation, (3)acid formation and (4)oil degradation products. In reality there is always a little of all the chemical stages occurring but on the whole additive depletion will start earlier than oxidation, which is earlier than acid formation and oil degradation products. You can also add a symptomatic 5th stage of lube degradation too. I will go through each of these in turn.
1. Additive depletion
All lubricating oils have additives designed to protect the lubricant from oxidative degradation and ultimately symptoms of failure including wear. These can include antioxidants including phenolic, amine and combination additives. Antiwear additive depletion is also a good measure of remaining active antiwear additives – this is different to the total of the zinc and phosphorus, as spent additive will still show as chemical elements in the sample elemental analysis. In engine oils there is also a Base Number additive designed to neutralise acids.
How is additive depletion tested?
In Engine oils the Base Number is the most widely tested additive. It is measured by testing how much acid needs to be added to consume all of the remaining alkali Base Number. This is a test performed routinely on all engine samples and you are all likely familiar with it if you have had an engine oil test.
Indirect antioxidant measurement – This was the original way to test for Antioxidants. Antioxidants are measured indirectly by a process called oxidation resistance or oxidation stability. These tests rely on accelerating the ageing of a lubricant and measuring how long it takes to deplete the additives so oxidation begins. A common version of this is the RPVOT or RBOT test that measures in minutes how long a lubricant antioxidant lasts in a rotating pressure vessel, also known as a bomb. These tests tend to be very expensive – maybe 20 times more expensive than a single oil sample and take a long time to run – perhaps a few days if the oil has a lot of antioxidants present. Hence this tends to be only used by a few select labs and usually only on extremely large turbine systems with big budgets and no requirement for fast results.
Antioxidant measurements can now be direct in recent years with measurements by e.g. the RULER test (amine and phenols) or OAL’s protective additive test suite (amine, phenol and phosphorus based anti-wear) now popular alternatives. These tests directly measure the antioxidants in the oil and can be compared to new oils to measure the level of depletion. They have the advantage they are fast and much less expensive than indirect methods. The RULER is sold as a field test, but its price point tends to be more at lab instrument prices – so I rarely see end users owning one and usually it is labs that have these. Also the reagent solution needed with each sample for RULER is about 3 to 4 times a normal oil analysis test price. This is cheaper and faster than RPVOT, but the price can still be prohibitive to most people and means the price the lab has to pass to the customer is high. The OAL protective suite is a lab only test but price wise is only a fraction of the alternatives and also covers antiwear additives too. Hence, this would be my go to option unless you have to use one of the other methods e.g. RPVOT to meet an OEM requirement.
An additional test that is often overlooked is colour. This does not directly measure antioxidants or even indirectly measure them, but as antioxidants are spent they can act as indicators (a little like litmus paper, which also contain a phenolic derivative to cause the colour change). This can happen at a localised level too so although all the additive is not spent or acids have formed a great increase in colour can be seen. A word of warning though, is that colour can change whilst there is still life in the oil hence a large colour change should be used as a trigger to have additional tests to determine the cause.
Now that we have covered additives the next point to consider is oxidation of the lubricant.
2. Oxidation
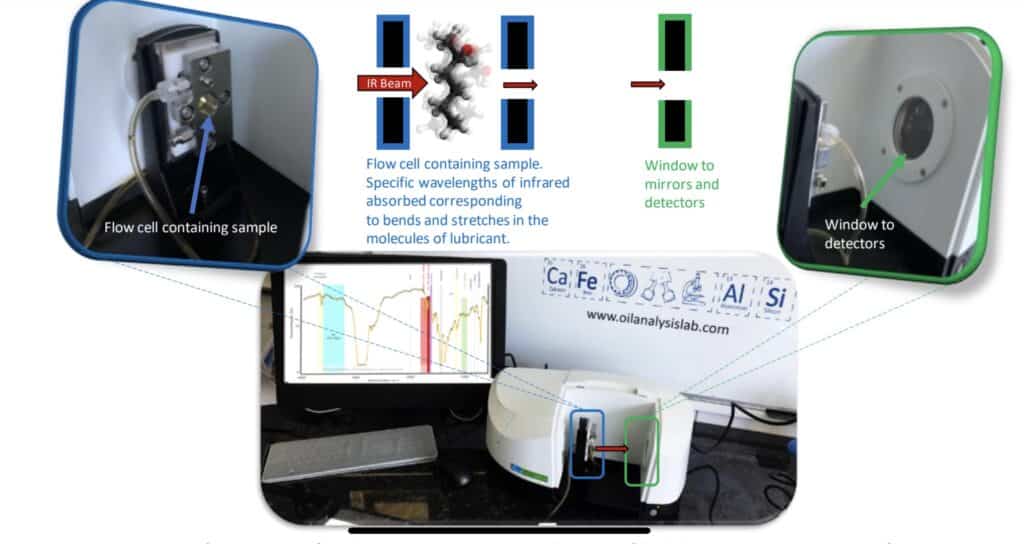
Oxidation is when an oil gains oxygen or in chemistry terms loses electrons. This can be seen by infra-red analysis where infra red light is shone through the sample and the various vibrations of the chemical compounds absorb some of the light that matches the frequency of the vibration. These vibrations have some funny names like rock, stretch, bends and even wag. A common chemical functional group called a carbonyl (C=O) vibration is measured as an indicator of the oxidation process. Carbonyls are seen in early degradation products of aldehydes (H-C=O).
Below are some example traces to illustrate oxidation. The graph is labelled with the main functional groups of interest. It is measured in transmittance so that means if all the light goes through the graph is at the top and if it is all absorbed it is at the bottom. So peaks point downwards.
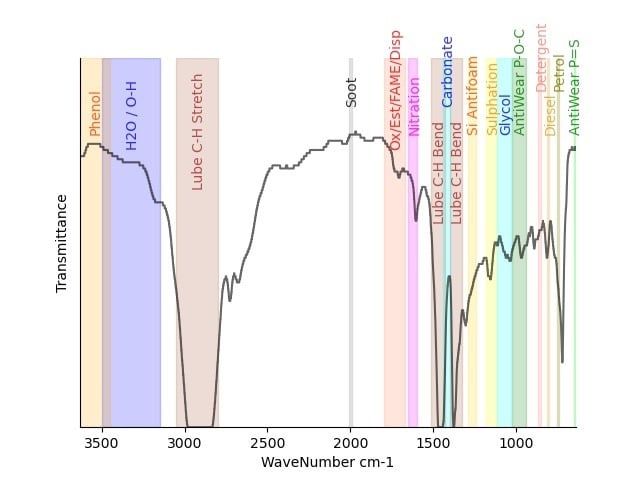
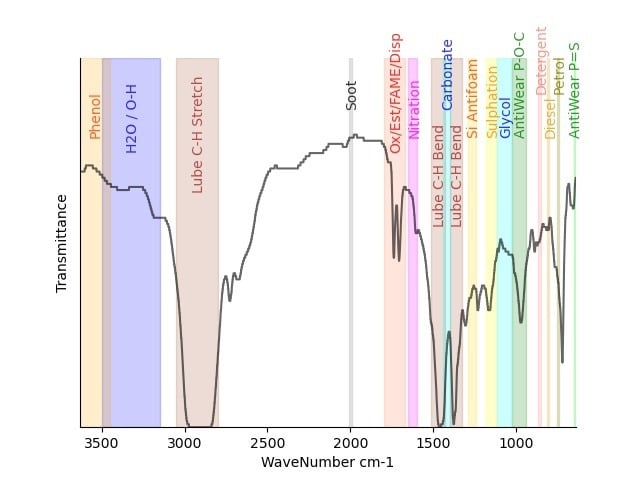
After oxidation to aldehydes, The carboxylic acids are the later degradation product (HO-C=O), still containing the carbonyl and still detectable by infrared, but these are when acids start to be detected too.
3. Acid formation

Once the oil has formed carboxylic acids this can be detected by Acid Number (AN), historically prefixed with a T for Total (TAN). This can be measured by titration with potassium hydroxide (KOH an alkali) and is measured in how much KOH is needed to neutralise the acid in a specific weight of sample. The units are hence mgKOH/g. Although the Base Number uses perchloric acid or Hydrochloric acid to titrate with the units (to allow easier comparison) are also used in mgKOH/g. Acid number can be measured by colour change – using a litmus type solution indicator that changes green from red when the alkali has neutralised the acid. On dark fluids like engine oils or lubricants that are dyed, this is not possible to run this test and hence a voltage based titration is performed instead called potentiometric titration. This is much less accurate than colour based tests, but sometimes has to be used if there is no alternative e.g. in engine oils.
Another test that can be used in combination with potentiometric acid numbers is measurement of initial pH to determine how acidic the oil is. The reason the test is called initial pH is pH is designed for aqueous (i.e. water based) samples. Obviously lube oils are not water based and hence can’t be measured. Instead the pH of the test solution containing the sample for an acid number is measured before titration and this is called the initial pH. This is very popular in gas engine analysis.
4. Oxidation degradation products
As the oil continues to oxidise,the process rapidly increases through the formation of free radicals. I won’t go into this in to too much detail, but if anyone is interested in free radical involvement in lubricant degradation, this is covered in my paid training courses. Drop a message on the contact us button below to find out more.
The end result of steps 1 to 3 is these oxidised and acidic materials form to such a concentration they can no longer be fully dissolved/suspended by the lubricant. These materials form sludges and varnishes. This can be observed by testing with an increased tendency for varnish potential. This tests how likely the oil is to form varnish particles. It is worth noting that this should be done before varnish has already started forming in the system to a serious extent to cause symptoms. This is because with severely oxidised oils the amount of varnish may be much higher than predicted by varnish potential testing because much of the varnish has already deposited and is no longer in the main circulating liquid. Hence filter analysis should be added rather than just lube varnish potential analysis to complement and catch these occurrences if suspected.
In addition to increased varnish potential, other physical properties may change such as viscosity which can increase in these situations. This is especially true of blends where the oil is made of multiple base oils – for instance if an ISO VG 46 has been made using an ISO VG 32 and an ISO VG 68. As the oil thermally degrades the thinner grade molecules termed the “light ends” volatilise off preferentially to the heavier thicker grade molecules bringing the oil closer to the thicker of the two grades. So in this example the ISO VG 46 as the ISO VG 32 volatilises off the oil becomes closer to the ISO VG 68. Although mixed blends such as this demonstrate the principle best, all non-synthetic lubricants will show this property to some extent as a lubricant is not one molecule type and is a mixture that the average just happens to be your viscosity grade.
Another thing to consider regarding viscosity increase is that this is true for mono-grade oils, but multi-grades you have two competing forces. For instance a 15W40 oil has additives in a SAE 15 to make it act like a 40 at high temperatures. This is achieved with viscosity index improvers. These include PIB – the stuff that makes some brands of cello-tape sticky. These additives curl up when cold, but when warm spread our and act a little like walking through a disco with your arms out to the side. You are likely to bump into more people with your arms out than by your side. You have increased the resistance to flow on the dance floor, which is what viscosity is defined as. Unfortunately, if you imagine you are now have your arms out in an engine where components are moving thousands of times a minute, moving your arms around like this sounds a bad idea and you are likely to lose one or two so that it’s almost as if you are walking with your arms by your side again (because you lost your arms). This is termed polymer shearing and can result in an oil no longer giving this increased viscosity and getting thinner with age. Hence in these situations as an oil additive is spent this can give a thinning, which may offset some of the thickening of the lubricant at first, but eventually the thickening process will win and the oil will sludge.
In hydraulic systems, varnish particles can also be observed in increased particle counts especially with the finer >4 and >6 micron sizes. Dilution particle counting does eliminate more of these softer particles when compared to neat testing, but I personally think you never remove them all, so particle counting should also be used to aid in spotting oxidation degradation products. As an aside, dilution counting is designed to remove soft particles such as additives and oxidation degradation products – ie non abrasive types of particles so the data can focus on the harder abrasive particles like dirt and wear. However, I don’t like the terms soft and hard particles as it suggests the soft particles don’t matter, when they can cause just as much problems as their hard counterparts, which we will discuss in the next section.
5. Symptoms
Symptoms are when the machinery starts to exhibit signs of oil degradation. This can include some physical properties such as colour change and viscosity changes as already mentioned. However it can also include sticking valves, blocked filters, overheating and wear. Oxidation products preferentially form in the hottest parts of the system, but their solubility decreases with temperature causing the material to deposit in cooling parts of the system. This insulates the system if deposition is in a cooler, and can rapidly lead to overheating and more varnish formation.
These oxidation products form thick sticky material, which is perfect for blocking an oil filter or sticking a valve. Although the materials are not themselves abrasive, they are poor lubricants and lead to increased wear too. Oil analysis and filter analysis can aid as late as step 5, but by this point is may be potentially too late.
Which is the best?
There is no best method. They all have their uses. Some can also be used in combination to give a better more rounded view of the lubricant. You need to factor in if you need every test and often general condition monitoring may not need any specific test just to check for oil degradation depending on your oil change frequency.
What I can say though is that for every £1 invested in intervening in stages 1 to 4 you can nominally save £10 in costs by preventing the oil reaching stage 5. Hence the importance of a regular analysis programme to assess the condition and highlight these problems early.
If you want to check the condition of your oil then please contact us by using the contact us button below.
